|
1 BaCl2 + 1 Double Displacement Na2SO4 □ 1 BaSO4 + 2 NaCl 1 Zn + 2 Single Displacement HCl □ 1 ZnCl2 + 1 H2į.  
2 KI + 1 Single Displacement Cl2 □ 2 KCl + I2ĭ. Already Balanced H2O + Synthesis SO3 □ H2SO4Ĭ. Already Balanced AgNO3 + Double Displacement KCl □ AgCl + KNO3ī. (Some equations may already be in balance.) In the space to the right, classify the reaction as a synthesis, decomposition, single replacement, or double replacement reaction.Ī. Practice: Balance each of the chemical equations below. Double replacementī- A compound reacts with an element to form a new compound & a different element. Single replacementĭ-Two compounds react to form two different compounds. DecompositionĬ-Two or more reactants combine to form one product. SynthesisĮ-A fuel is combined with oxygen to produce carbon dioxide and water. Using the chemical equations in the gizmo as a guide, match the following definitions to the type of reaction:Ī-One reactant is broken down into two or more products. Match: Most chemical reactions can be classified as one of four types. Question: How are chemical reactions classified? A compound is a substance made of more than one kind of atom, such as water (H2O) or table salt (NaCl). An element is a substance consisting of one kind of atom, such as aluminum (Al) or oxygen gas (O2). Introduction: Chemical equations show how compounds and elements react with one another. (These equations are not in the Gizmo.)īalancing Chemical Equations Gizmo Answers – Activity B Practice: Balance the following chemical equations. Check your answers and then write the balanced equations: Use the Choose reaction drop-down menu to see other equations, and balance them. 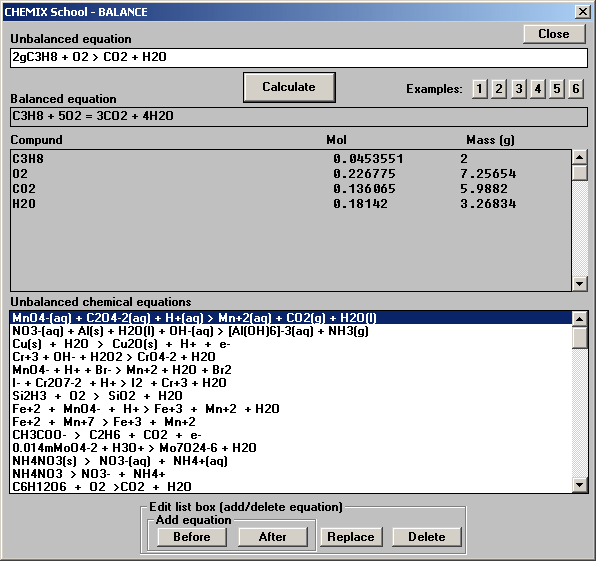
When you are done, turn on Show summary to check your answer. In the Gizmo, use the up and down arrows to adjust the numbers of hydrogen, oxygen, and water molecules until the equation is balanced. The equation is balanced when there are equal numbers of each type of atom represented on each side of the equation. Question: How are chemical equations balanced? To balance the equation, you cannot change the structure of any of the molecules, but you can change the number of molecules that are used. Introduction: The equation H2 + O2 □ H2O is unbalanced because there are two oxygen atoms on the reactant’s side of the equation and only one on the product’s side of the equation. Is this reaction balanced?Īns: No, because there are two oxygen atoms on the reactant’s side of the equation and only one product side of the equation.īalancing Chemical Equations Gizmo Answers – Activity A A chemical equation is balanced if the number of each type of atom on the left side is equal to the number of each type on the right side. In general, what does a subscript (such as the “2” in H2) tell you about the molecule?Īns: The subscript means the number of atoms of each element is in the molecule. How many hydrogen and oxygen atoms are in a water molecule (H2O)? How many atoms are in an oxygen molecule (O2)? How many atoms are in a hydrogen molecule (H2)? In the Balancing Chemical Equations Gizmo, look at the floating molecules below the initial reaction: H2 + O2 □ H2O. This process is summarized by a chemical equation. In a chemical reaction, reactants interact to form products. Free chemical equation balancer full#Assuming a s’more requires two graham crackers, one marshmallow, and one piece of chocolate, how many smores could you make with the ingredients shown?Īns: With this assumption and the ingredients shown, only 2 full smores could be made. Also, 4 smores would require 8 graham crackers but there are only 5 crackers shown. Despite there only being 3 chocolate bars, 4 smores were made. Introduction: The scouts are making s’mores out of toasted marshmallows, chocolate, and graham crackers.Īns: Altogether it equals 4 smores however the quantity of the marshmallows, chocolate, and gram crackers are not the same. Vocabulary: coefficient, combustion, compound, decomposition, double replacement, element, molecule, product, reactant, single replacement, subscript, synthesis NOTE: All answers are checked twice before publishing them to you.
0 Comments
Leave a Reply. |
AuthorChristine ArchivesCategories |
- Azure storage emulator opends-dll
- Blog
- Dragon quest 3 snes version
- Mkv video player reddit
- Story of talaash movie 2012
- Smart kapp app store
- Geektyper matrix
- Epson l210 resetter program
- Eminem without me download free mp3
- Text to speech voices vista
- Microsoft 3d movie maker
- Download max payne 3 blackbox
- Imperium galactica 2 widescreen
- Grabber atx
- One piece 935
- Office 2016 activator and key
- How to play terraria 1-1 on pc
- Secret of mana rom version check
- Softraid m1


 RSS Feed
RSS Feed
